This past week the Wall Street Journal printed an article following the announcement by Health Secretary Kennedy that the FDA was going to lift restrictions on peptide treatments. We are noting a rising of patient self-reporting consumption of peptide therapy protocols. Many of these patients are doing this in addition to paying us for comprehensive medical access and care. At this point, I felt it is time for my own self education and to post to the public on this topic.
Interestingly, despite Americans shouting that health care is unaffordable, it appears a big chunk of people are willing to shell out big bucks to keep up with the Jones’. Keep this in mind the next time your neighbor or a stranger tells you they can’t afford health care.
When you combine peptides and hormone therapy, you’re no longer looking at fringe medicine—you’re looking at a multi-billion-dollar, cash-pay health economy operating in plain sight, largely outside the traditional health care access point, namely doctors.
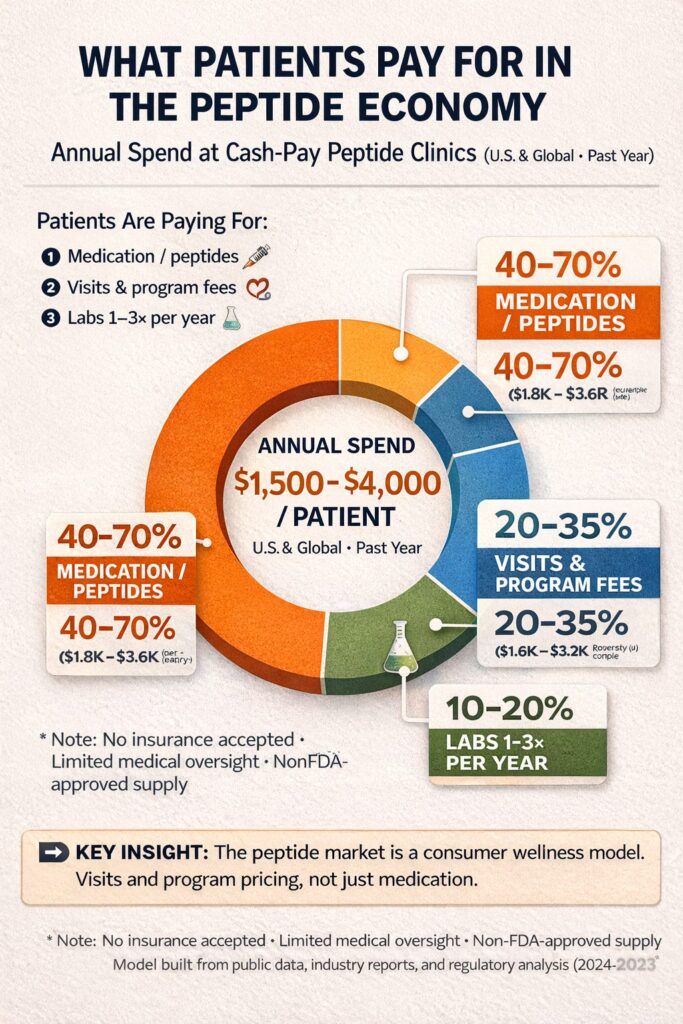
And if we add the gateway to Peptide therapy, namely, alternative hormone replacement therapy we see an even larger spend going on…

Globally this market may be 30 billion dollars or more.

It makes sense to me that Secretary Kennedy would loosen restrictions on peptides. He gets to poke big pharma in the eye for starters. The COVID 19 pandemic provided the opportunity for loosened compound pharmacy regulation. Big pharma couldn’t meet market demand for Ozempic and Mounjaro. Shortages are a scenario where patent law can be suspended and President Biden’s administration did just that. The result was a rapid response “black/gray market for the generic/compounded versions of Ozempic and Mounjaro. These patent protected prescriptions aren’t typical pharma chemical molecules. They are, in fact… peptides called Glp-1 agonists. Actually these compounds make up the Lion’s share of the current US and likely global peptide market.
Noting the success of these scientifically studied and proven human peptide analogues (Glp1 agonists), I am learning that the telehealth supply chain and the non-physician hormone and aesthetic store fronts are pitching unproven but theoretically plausible peptide stacks onto patients. All of them promise leaner body fat and improved muscle and ligament healing. A more powerful and youthful YOU so to speak. Objectively the results can all be explained by the Glp-agonists but hey, why not jab yourself with some more products and get even “better” outcomes.
As a physician, I am chagrined as well as concerned about health extenders opening infusion clinics, pumping vitamins, amino acids, and who knows for sure what all into hangover victims and folks that just want to feel better.
Medicine is not just about prescribing pills; it is about pattern recognition. With over 30 years of clinical experience, I view myself not just as a physician, but as a medical detective.
When I consult with a patient seeking a second opinion—whether they are driving up from Naples, coming from Sanibel, or flying in for the season—I am looking for the subtle clues that others miss.
The GLP-1 compounding boom demonstrated that patients are increasingly use alternative, consumerized care pathways when conventional channels are slow or expensive. Telehealth, cash pay, and compounding pharmacies functioned as a scalable distribution system for GLP-1s, filling a supply and access gap for a blockbuster therapy. These channels did more than fill a temporary gap: they demonstrated that patients will route around insurer-mediated pathways when access is slow, coverage is uncertain, or prices are high. In doing so, these pathways expanded drug access and customization, but often with less oversight and frequent conflicts with patent protections.
The growth of compounding also highlighted a longstanding tension in biopharma: patent exclusivity is designed to reward costly R&D, yet high prices and uneven coverage can create strong pressure for workarounds. Compounded GLP-1s exposed how quickly those workarounds can now scale. This pattern will likely be reproduced with future high-demand therapies, forcing regulators, manufacturers, and clinicians to balance access, safety, and incentives for innovation as more care moves outside traditional clinical settings and insurer-mediated systems.
Peptides (Non-Biologics): Any polymer composed of 40 or fewer amino acids is generally classified as a drug (small molecule). These fall under the Hatch-Waxman Act for patenting and generic approvals. The first patented therapeutic peptide is insulin.
Historical Context
Defining the Class: Insulin was the first substance to prove that a peptide (a chain of amino acids) could serve as a life-saving drug. It is a 51-amino acid peptide, though modern regulatory definitions (like the Hatch-Waxman Act mentioned in your draft) sometimes distinguish between “small” peptides (under 40 amino acids) and larger proteins. The law changed in 2020 regarding peptide definition. This is why insulin is still considered the first peptide despite having 51 amino acids. So, legally speaking, the FDA now classifies insulin as a protein/biologic, not a peptide. This transition was designed to open the door for biosimilars (the biologic version of generics).
First Synthetic Versions: Decades later, insulin continued its “firsts” by becoming the first recombinant DNA (biotechnological) drug approved by the FDA in 1982 (Humulin).
There are many peptides that are prescription medications. Examples include the Glp-1 agonist peptides. Namely, liraglutide, semaglutide, tirzepatide. Liraglutide is a daily injection now available generically. The other two are the name brand products we all know and love which are also currently still being offered in compounding pharmacies. Other examples include teriparatide (Forteo) used for osteoporosis treatment, desmopressin (DDAVP) used for diabetes insipidus and bedwetting, and bremelanotide (Vyleesi) for female sexual dysfunction.
As of late March 2026, the specific number of peptides moving to Category 1 is 14, according to the directive from Health and Human Services (HHS) Secretary Robert F. Kennedy Jr. Category one allows legal marketing and preparation and are considered posing no significant safety risks.
On February 27, 2026, Secretary Kennedy announced a major policy shift to “end the war on peptides” by reclassifying approximately 14 of the 19 peptides that the FDA had previously restricted in late 2023. 1
Following the landmark February 2026 announcement regarding the reclassification of therapeutic compounds, several “trendy” peptides are making a high-profile return to compounding pharmacies. These options have become particularly popular in anti-aging and recovery protocols:
BPC-157 (Healing & Recovery): Often called the “Body Protection Compound,” this peptide is a favorite for systemic repair, specifically targeting gut health and the accelerated healing of tendons and ligaments.
GHK-Cu (Skin & Anti-Aging): A naturally occurring copper peptide frequently used in both premium serums and injections to stimulate collagen production and visibly reduce wrinkles.
AOD-9604 (Targeted Fat Loss): This fragment of Human Growth Hormone is designed to trigger lipolysis (fat burning) without the unwanted side effect of affecting blood sugar levels.
MOTS-c (Metabolic Health): Widely referred to as an “exercise mimetic,” it is prized for its ability to boost mitochondrial function and overall cellular energy.
CJC-1295 / Ipamorelin (Growth Hormone Support): Typically administered as a synergistic “stack,” these peptides stimulate the body’s natural growth hormone production to improve deep sleep and support muscle maintenance.
Patients are coming in telling me they are getting the Wolverine stack. I am thinking “what???”.
The “Wolverine Stack” (or Wolverine Blend) is a popular nickname for the combination of two specific regenerative peptides: BPC-157 and TB-500 (Thymosin Beta-4). It is named after the Marvel character because of the intended effect of “superhuman” healing and tissue repair.
Disclaimer: This information is for educational purposes only and is not intended to replace professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
Get expert analysis from one of Southwest Florida’s most experienced Internal Medicine and Lipidology specialists. Call us today to discuss your case.

Southwest Florida’s premier Medical Home. Dedicated to prevention, access, and the patient relationship. Serving Fort Myers, Sanibel, Cape Coral, Bonita Springs, Naples & surrounding Southwest Florida areas.
Contact
6160 Winkler Road
Fort Myers, FL 33919
(239) 362-3005
Fax: (239) 362-3392
staff@imlwp.com